|
Case Series
The use of bioactive glass as a posterolateral graft in patients with infected spondylodesis
1 Doctor of Medicine, MD, Orthopedics, Maastricht University Medical Centre, Maastricht, The Netherlands
2 Orthopedic Surgeon, MD, PhD, Orthopedics, Maastricht University Medical Centre, Maastricht, The Netherlands
Address correspondence to:
Paulien van de Voorde
Postbus 5800, 6202 AZ Maastricht,
The Netherlands
Message to Corresponding Author
Article ID: 100019Z14PV2019
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
van de Voorde P, Geurts JAP. The use of bioactive glass as a posterolateral graft in patients with infected spondylodesis. J Case Rep Images Orthop Rheum 2019;4:100019Z14PV2019.ABSTRACT
Introduction: Bioactive glass is used increasingly, also as a bone graft in spondylodesis. Autologous bone is used for the pursuit of a solid spinal fusion in spondylodesis. Iliac crest bone is most commonly used as a donor site for obtaining autologous bone graft. The use of autologous bone is associated with some risks and higher comorbidity. For these reasons, surrogates are used. In the case of infected spondylodesis material, in contrast to infection treatment with arthroplasty, the material remains in situ to maintain stabilization. In such cases, only the autologous bone grafts are removed. Therefore, a substitution material will have to be put back that generates fusion and offers treatment to the underlying infection. Bioactive glass could potentially make an important contribution to this.
Case Series: In this case series, we want to provide an initial insight into the use of bioactive glass, used in a combined setting, in patients with infected spondylodesis material. We present four cases of different patients.
Conclusion: In all cases, there were no signs of persisting infection and it was confirmed that the use of bioactive glass also maintains the spinal fusion.
Keywords: Bioactive glass, Infection, Spondylodesis
Introduction
The use of bioactive glass (S53P4) (by BonAlive Biomaterials Ltd., Turku, Finland) is increasing. Where it was initially used primarily for the treatment of osteomyelitis, as a treatment for bone and bone marrow infection, it is also used as a bone graft substitute in spondylodesis. In the classical procedure of spondylodesis, in addition to the osteosynthesis material, autologous bone is used for the pursuit of a solid spinal fusion. This working method is considered as a gold standard [1],[2]. The most commonly used donor site for obtaining autologous bone graft is from the iliac crest. This acquired bone is attached to the autologous bone that has already been obtained, which becomes available during the removal of the processus spinosi, processus transversi, and facet joints. The use of autologous bone is associated with a longer duration of surgery, increased blood loss, but also with the morbidity that the patient may have depending on the donor site [3],[4]. The availability of spongy bone is also finite. For these reasons, surrogates are used. In addition to the use of autologous bone grafts, various substitution materials are used in orthopedics, including, for example, allografts, beta-tricalcium phosphate, coralline hydroxyapatite, but also bioactive glass [5],[6],[7],[8]. Previous animal research and research in periodontology have already demonstrated the effect in spinal fusion when bioactive glass is used as a surrogate in addition to the autologous bone graft. In their research, Frantzén et al. describe that bioactive glass can be considered as a good alternative as bone graft [9]. In this study, bioactive glass was used on one side of the spine as an addition to bone graft on the contralateral side. Radiological fusion, as taken by computed tomography (CT) scans, was evaluated and assessed for the autologous bone graft side and the bioactive glass side. In this study, a fusion rate of 100% was observed for autologous bone graft as detected by CT. On the bioactive glass side, a solid fusion of 80.5% was observed. In a prospective study with 10 years of follow-up, Rantakokko et al. showed that bioactive glass is not only a good alternative, but at the same time, it is also a safe supplement that can be used in spondylodesis surgery in patients with unstable lumbar fractures [10]. Research on the use of bioactive glass in patients who underwent spondylodesis is still too limited to be able to judge whether bioactive glass can be used on its own as a bone graft. Nowadays it is only used in combination with autologous bone. Bioactive glass is an osteostimulative and osteoconductive material. Extensive research into the different effects of bioactive glass has taught us that bioactive glass also leads to a reduction in inflammatory responses [11],[12]. The study by Frantzén et al. also does not describe postoperative wound infections [9]. If infected spondylodesis material is involved, this should, in contrast to infection treatment within arthroplasty, remain in situ to maintain stabilization. In such cases, it is only possible that autologous bone grafts are removed. Therefore, a substitution material will have to act as a replacement that generates fusion and offers treatment to the underlying infection. Bioactive glass could potentially make an important contribution to this.
Research in this combined setting, with the use of bioactive glass, in patients with infected spondylodesis material, has not been investigated before. With the following case presentations from patients, we want to provide an initial insight here.
CASE SERIES
Case 1
In the first case, we present a 70-year-old patient who underwent the first operation in a hospital elsewhere. Patient characteristics are shown in Table 1. Initially, a decompression of L5 and anterior spondylodesis was performed elsewhere at the L5-S1 level in 2001. Subsequently, this level was expanded to L4-S1, with a spinal stenosis L4-L5 (2013) and then to L3-S1 with spinal stenosis at level L3-L4 (2016). In the latter case, there was an increase in pain on the third day postoperatively, with mobilization becoming more and more difficult. There was also discharge from the wound hematoma with subsequent wound leakage. As a result, wound cultures were taken. These cultures were found to be positive with Escherichia coli, Staphylococcus aureus, Proteus mirabilis, and Corynebacterium. Blood cultures were also taken during the development of a fever. The patient underwent surgical debridement in the development of sepsis. Deep and superficial cultures were taken perioperatively. This procedure was then repeated twice in case of persistent fever and increased infection parameters. After the first debridement, postoperatively, long-term antibiotic treatment was started. Initially, intravenous treatment with Flucloxacillin (6 dd 1000 mg) was started and switched to Amoxicillin (4 dd 1000 mg) on positive cultures with E. coli. A one-time gift of Gentamycin (500 mg) was administered on suspicion of septic profile. At a second debridement, Gentamycin beads were left behind (120 beads in depth and 60 in intermediate layers). Postoperatively, intravenous treatment with Cefazoline (3 dd 1000 mg) was initiated and Rifampicin was started on a dry wound (2 dd 450 mg). The Gentamycin beads were removed again at a third debridement. The Cefazoline and Rifampicin, as described above, were continued. With the completion of intravenous treatment over a period of two weeks, a switch was made to oral therapy with Cotrimoxazole (2 dd 960 mg) in combination with the Rifampicin. Given the presence of osteosynthesis material, the patient was treated with antibiotics for a total of three months. The infection parameters normalized [sedimentation (BSE) 42 mm/hour, hemoglobin (Hb) 6.7 mmol/L, leukocytes (L) 6.2 × 109 mmol/L, c-reactive protein (CRP) 9 mg/L].
In 2018 the patient presented with a pseudoarthrosis at level L3-L4 (Table 2). A spondylodesis of level L3-S1 was performed with 10 cc bioactive glass. Pre- and perioperatively the patient was treated with Cefazoline, with a single gift of Cefazoline (2 g) preoperatively and postoperatively Cefazoline prophylaxis for 24 hours (3 dd 1000 mg). There was no fever after surgery. There was wound healing (dry wound) on the second day postoperatively and a good position of the osteosynthesis material was seen during X-ray examination. The patient left the hospital postoperatively on the third day.
The patient was seen again three months postoperatively at the Orthopedics outpatient clinic. He was satisfied and no longer experienced pain. A good fixation and position of the spondylodesis L3-S1 were observed. There was only the presence of muscle-related complaints about the thoracic level.
Case 2
The second case concerns a 69-year-old patient who was previously operated twice by neurosurgery. Patient characteristics are shown in Table 1. Initially, interlaminar L2-L3 decompression was performed in case of neurogenic claudication with neurological failure (2014). With stenosis on L4-L5 a laminectomy and discectomy of level L4 and L4-L5 were performed (2015). Posterolateral spondylodesis was performed from levels L3 to S1, with decompression and foraminotomies at levels L3-L4, L4-L5, and L5-S1 (2016). There was wound healing on the tenth day postoperatively. Three days later, the patient went home in good clinical condition.
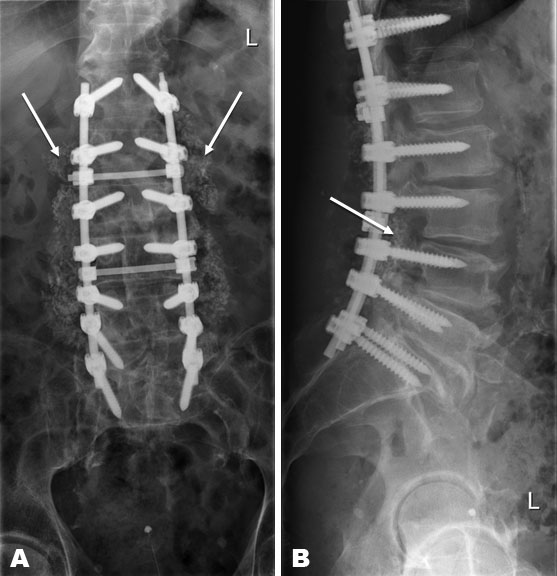
In the short term, five days after the discharge from the previous admission, the patient was readmitted with an increase in back pain, fever (temperature above 38.5°C) and increased infection parameters (sedimentation unknown, Hb 7.4 mmol/L, L 21.4 × 109 mmol/L, CRP 326 mg/L). In suspicion of deep infection, the patient underwent debridement with leaving behind 180 Gentamycin beads (3 times 60 beads). Cultures were taken perioperatively. Postoperatively, intravenous treatment with Cefazoline (3 dd 1000 mg) was started. Based on positive cultures (Staphylococcus epidermidis), Vancomycin was started intravenously based on blood levels, given resistance to Flucloxacillin and Cefazoline. In addition, Rifampicin (2 dd 450 mg) was started with a dry wound. Under antibiotic treatment, the infection parameters decreased steadily. However, a revision operation (Table 2) was performed in the event of sustained wound leakage and in situ beads. A debridement was carried out with the removal of the Gentamycin beads and again with the cultivation of cultures. The previous spondylodesis was extended to level Th12 (Th12-S1) with a 20 cc bioactive glass (2 × 10 cc). The patient recovered quickly postoperatively. Wound healing occurred on day 17. X-ray control showed a good position and fixation of the material (Figure 1A and Figure 1B). With antibiotic treatment, the infection parameters decreased. In clinically good condition, the patient was discharged with oral antibiotics, Minocycline (2 dd 100 mg) and Rifampicin (2 dd 450 mg). In total, the patient was treated for three months due to the presence of osteosynthesis material in the body.
Postoperatively, the course was complicated by a lacunar infarction in the right hemisphere with hypertonic paresis of the left arm and leg. A partial spinal cord injury based on a spinal canal stenosis C3-C4 in combination with a fall on the back of the head was associated with compressive myelopathy with surgical recovery. Due to the described postoperative course, the outpatient followup was not entirely satisfactory. At the first outpatient check-up, three months postoperatively (Figure 2A and Figure 2B), the patient indicated that his back problems had not improved and even increased compared to the preoperative situation. Further outpatient follow-up is missing.
Case 3
The third case describes a 56-year-old patient. Patient characteristics are shown in Table 1. Initially, the patient presented with high-grade spinal stenosis on levels L2-L3 and L3-L4. Operative decompression laminectomy L3-L4 was performed for a herniated nucleus pulposus (HNP) L3-L4 with nerve compression. Fusion was created from levels L2 to L5. Intracorporal cages were placed on level L4-L5 (2017) in case of recesses stenosis L4-L5 on the right side, with compression L5 and right intraforaminal HNP with compression of L4. Preoperatively, the patient received Cefazoline (2 g) with postoperative Cefazoline prophylaxis 24 hours (3 dd 1000 mg). In the case of evident puss (wound) on the 13th postoperative day, in combination with increased infection parameters (sedimentation unknown, L 11.4 × 109 mmol/L, CRP 417 mg/L) and temperature 37.8°C, it was decided to perform surgery. Debridement was performed by placing Gentamycin beads. Postoperatively, intravenous antibiotics were started with Augmentin (4 dd 1200 mg). Based on positive cultures (S. aureus), a switch was made to Flucloxacillin (6 dd 1000 mg) and Rifampicin was started orally (2 dd 450 mg) on a dry wound. Among antibiotics, the patient recovered quickly and a downward trend was observed in the infection parameters. An oblique fracture of corpus L5 was seen on X-ray control. In a renewed operation (Table 2), the previous spondylodesis was extended to level S1 for this, the beads were removed and bioactive glass (10 cc) was placed posterolaterally. New cultures were taken perioperatively. These cultures turned out to be negative. Postoperatively, Flucloxacillin intravenously and Rifampicin were continued orally. Intravenous antibiotics were switched to oral antibiotics after two weeks with Flucloxacillin and Rifampicin. With fever and increasing infection parameters, therapy was switch back to intravenous therapy with Flucloxacillin for three months in total. Afterward, there was no longer a fever. A good position and fixation of the osteosynthesis material were seen. Wound healing was achieved on the fifth day postoperatively. The patient left the hospital in good clinical condition, after being admitted for three weeks, home with intravenous antibiotics.
The patient was followed outpatient with long-term use of intravenous antibiotics. No wound problems occurred. The infection parameters decreased steadily. After a period of five months postoperatively, the patient developed an increase in back pain and spasticity of the right leg. In additional examinations, a pseudoarthrosis of the L5-S1 level was seen with the outbreak of screw S1 on the right. The patient was assisted in this by performing surgery with the expansion of spondylodesis to iliac level (L2-iliac) with decompression of L5 bilaterally and placing a new S1 screw on the right. The patient was seen again six weeks later at the Orthopedics outpatient clinic. She was complaint free and felt reborn. A good position and fixation of the spondylodesis material were achieved. In addition, there was complete wound healing.
Case 4
The fourth and final case concerns a 19-year-old patient with neuromuscular scoliosis for which she underwent surgical correction with dorsal spondylodesis of the Th2 to S1 level. Patient characteristics are shown in Table 1. Preoperatively, Cefuroxime (1500 mg) was administered as a prophylactic treatment. Postoperatively, the patient was treated with Cefuroxime prophylaxis for 24 hours (3 dd 1500 mg). There was wound healing on the sixth day postoperatively, after which the patient left the hospital on the seventh day.
Ten days after the discharge from the hospital, the patient reported back to the hospital with wound leakage, in the case of wound dehiscence at the distal part of the wound. A slight increase was seen in infection parameters (sedimentation 55 mm/hour, Hb 5.7 mmol/L, L 7.7 × 109 mmol/L, and CRP 45 mg/L). There was no fever. With regard to the wound leakage and increased parameters, a debridement was performed with the placing of Gentamycin beads. Based on positive perioperative cultures, with E. coli, treatment with Cefuroxime intravenously (3 dd 1500 mg) was targeted. This antibiotic treatment was continued until the next surgical procedure (Table 2) in which the beads were removed and 10 cc bioglass was left behind. Postoperatively, the treatment was continued with intravenous treatment with Cefuroxime (3 dd 1500 mg) for two weeks. Perioperative cultures showed no growth. In total, antibiotics were treated for three months through oral therapy with Cotrimoxazole (2 dd 960 mg). Wound healing occurred from the fourth day postoperatively. The infection parameters declined steadily with antibiotic treatment. At discharge, on day ten postoperatively, the values were normalized (sedimentation 20 mm/hour, L 3.2 × 109 mmol/L, and CRP 10 mg/L). A year postoperatively there was a stable situation, the pain was sustainable and mobilization was performed with a walker.
Discussion
Bioactive glass (by BonAlive Biomaterials Ltd., Turku, Finland) is an osteostimulative, osteoconductive, and antibacterial bone substitute [13]. It forms a chemical bond with hydroxyapatite and provides a scaffold for the formation of new bone [5]. Bioactive glass dissolves over time and is replaced by new bone. Composition of the glass (e.g., S53P4, 53% SiO2, 23% Na2O, 20% CaO, 4% P2O5), granule size and granule roughness are all important factors contributing to the cell and extracellular tissue reactions observed for bioactive glass [14]. Bioactive glass gained European approval for orthopedic use as a bone graft substitute in 2006. No adverse effects of using bioactive glass have been described. Extensive research into different effects of bioactive glass shows that bioactive glass also leads to a reduction in inflammatory responses. The study by Frantzén et al. also does not describe postoperative wound infections, in the use of bioactive glass [9].
With the knowledge of the use of bioactive glass in the treatment of bone infections and the use of bioactive glass as a possible supplement to the use of autologous bone grafts, this was used and described herein a combined setting. In every case described there was a (wound) infection with underlying infected osteosynthesis material. Several debridements were performed, each time with taking extensive tissue sampling. In three of four cases, it was decided to apply polymethyl methacrylate (PMMA) beads with Gentamycin. Postoperatively, intravenous treatment with antibiotics was started, which was adjusted according to cultures. If PMMA beads were placed, they were also removed in the latter instance. In this procedure, a debridement was still performed and before the end of the operation, with the closure of the different layers of the skin, bioactive glass (10 or 20 cc) was left as a posterolateral graft. Intravenous antibiotics were continued for two weeks. If wound healing occurred and a downward trend was observed in the infection parameters, patients were discharged from the hospital with oral antibiotics for the total duration of three months. In all cases, the underlying infection was cured and spinal fusion was established and confirmed by X-ray examinations.
In contrast to the treatment of infected knee or hip arthroplasty, there are no clear guidelines for the treatment of infected spondylodesis. If there is an infection in spondylodesis, removal of the osteosynthesis material in toto is not an option. This is because of the need of maintaining the acquired stabilization. In that case, it is extremely important to remove as much infected tissue as possible, in addition to the autologous bone gaps, and to replace it with a substitution material that provides fusion and treatment to the underlying infection.
With spondylodesis, it is important to make a distinction between an early, latent, or late infection. A distinction in localization of infection is also important [15]. If there is superficial wound infection, treatment can be started with intravenous antibiotics, possibly in combination with superficial incision and drainage. If there is infection below the fascia, a single treatment with antibiotics is usually insufficient and extensive debridement must be performed (DAIR). Any abscesses must be drained. In the case of an immune-compromised patient or unidentified bacterium, multiple debridements may be necessary to completely eradicate the bacterium. If the osteosynthesis material is loose perioperatively, this must be removed and replaced. The other essential material must remain in situ to avoid instability or the occurrence of deformity. If several operations are performed, the recommendation is made to leave PMMA beads, which release a high concentration of antibiotics at the wound infection site [15]. When performing multiple debridements, the involvement of a plastic surgeon may also be necessary for optimal wound treatment [16],[17].
Conducting a debridement is always combined with extensive antibiotic treatment. This usually involves a combination of intravenous treatment for two weeks followed by ten weeks of oral therapy, based on the culture results. The total duration of treatment is three months, given the retention and presence of (infected) osteosynthesis material. Multiple studies show that the debridement, antibiotics, and implant retention (DAIR) protocol is effective [18]. If this is done postoperatively within three months, this is considered to be the most effective [19]. Research also shows that when a late infection occurs, preference is given to remove the osteosynthesis material. This is partly due to the development of a biofilm [20]. Although in the case of spinal surgery the removal of the osteosynthesis material may have various side effects, such as spinal instability, radicular pain, and back pain [21]. This means that both goals are opposite of each other.
Despite extensive research already described, there remains uncertainty and there is no clear protocol with regard to the treatment of infected spondylodesis. Additional and ongoing investigations are still looking for the best possible treatment for eradicating the infection if the osteosynthesis material remains in situ. This with an aggressive debridement, possibly in combination with vacuum drainage (closed or open system with drain placement), primary wound closure and antibiotic treatment during a standard period.
Conclusion
Full wound healing was achieved and infection parameters normalized with the used antibiotic policy. No side effects or adverse effects have been described with the use of bioactive glass. In conclusion, both predefined goals have been achieved, in absolutely three of the four cases. Seen after the completed treatment process, with the preservation of the infected spondylodesis, there was no question of continuous infection and it was confirmed that the use of bioactive glass also maintains the spinal fusion.
REFERENCES
1.
2.
Korovessis P, Koureas G, Zacharatos S, Papazisis Z, Lambiris E. Correlative radiological, self-assessment and clinical analysis of evolution in instrumented dorsal and lateral fusion for degenerative lumbar spine disease. Autograft versus coralline hydroxyapatite. Eur Spine J 2005;14(7):630–8. [CrossRef]
[Pubmed]

3.
Robertson PA, Wray AC. Natural history of posterior iliac crest bone graft donation for spinal surgery: A prospective analysis of morbidity. Spine (Phila Pa 1976) 2001;26(13):1473–6. [CrossRef]
[Pubmed]

4.
Banwart JC, Asher MA, Hassanein RS. Iliac crest bone graft harvest donor site morbidity. A statistical evaluation. Spine (Phila Pa 1976) 1995;20(9):1055–60. [CrossRef]
[Pubmed]

5.
Acharya NK, Kumar RJ, Varma HK, Menon VK. Hydroxyapatite-bioactive glass ceramic composite as stand-alone graft substitute for posterolateral fusion of lumbar spine: A prospective, matched, and controlled study. J Spinal Disord Tech 2008;21(2):106–11. [CrossRef]
[Pubmed]

6.
Miyazaki M, Tsumura H, Wang JC, Alanay A. An update on bone substitutes for spinal fusion. Eur Spine J 2009;18(6):783–99. [CrossRef]
[Pubmed]

7.
Fujibayashi S, Shikata J, Tanaka C, Matsushita M, Nakamura T. Lumbar posterolateral fusion with biphasic calcium phosphate ceramic. J Spinal Disord 2001;14(3):214–21.
[Pubmed]

8.
Chen WJ, Tsai TT, Chen LH, et al. The fusion rate of calcium sulfate with local autograft bone compared with autologous iliac bone graft for instrumented short-segment spinal fusion. Spine (Phila Pa 1976) 2005;30(20):2293–7. [CrossRef]
[Pubmed]

9.
Frantzén J, Rantakokko J, Aro HT, et al. Instrumented spondylodesis in degenerative spondylolisthesis with bioactive glass and autologous bone: A prospective 11-year follow-up. J Spinal Disord Tech 2011;24(7):455–61. [CrossRef]
[Pubmed]

10.
Rantakokko J, Frantzén JP, Heinänen J, et al. Posterolateral spondylodesis using bioactive glass S53P4 and autogenous bone in instrumented unstable lumbar spine burst fractures. A prospective 10-year follow-up study. Scand J Surg 2012;101(1):66–71. [CrossRef]
[Pubmed]

11.
Vrouwenvelder WC, Groot CG, de Groot K. Histological and biochemical evaluation of osteoblasts cultured on bioactive glass, hydroxylapatite, titanium alloy, and stainless steel. J Biomed Mater Res 1993;27(4):465–75. [CrossRef]
[Pubmed]

12.
Wilson J, Pigott GH, Schoen FJ, Hench LL. Toxicology and biocompatibility of bioglasses. J Biomed Mater Res 1981;15(6):805–17. [CrossRef]
[Pubmed]

13.
Lindfors NC, Hyvönen P, Nyyssönen M, et al. Bioactive glass S53P4 as bone graft substitute in treatment of osteomyelitis. Bone 2010;47(2):212–8. [CrossRef]
[Pubmed]

14.
Välimäki VV, Moritz N, Yrjans JJ, Dalstra M, Aro HT. Peripheral quantitative computed tomography in evaluation of bioactive glass incorporation with bone. Biomaterials 2005;26(33):6693–703. [CrossRef]
[Pubmed]

15.
Hegde V, Meredith DS, Kepler CK, Huang RC. Management of postoperative spinal infections. World J Orthop 2012;3(11):182–9. [CrossRef]
[Pubmed]

16.
Mitra A, Mitra A, Harlin S. Treatment of massive thoracolumbar wounds and vertebral osteomyelitis following scoliosis surgery. Plast Reconstr Surg 2004;113(1):206–13. [CrossRef]
[Pubmed]

17.
Dumanian GA, Ondra SL, Liu J, Schafer MF, Chao JD. Muscle flap salvage of spine wounds with soft tissue defects or infection. Spine (Phila Pa 1976) 2003;28(11):1203–11. [CrossRef]
[Pubmed]

18.
Janssen DMC, Kramer M, Geurts J, Rhijn LV, Walenkamp GHIM, Willems PC. A retrospective analysis of deep surgical site infection treatment after instrumented spinal fusion with the use of supplementary local antibiotic carriers. J Bone Jt Infect 2018;3(2):94–103. [CrossRef]
[Pubmed]

19.
Manet R, Ferry T Castelain JE, et al. Relevance of modified debridement-irrigation, antibiotic therapy and implant retention protocol for the management of surgical site infections: A series of 1694 instrumented spinal surgery. J Bone Jt Infect 2018;3(5):266–72. [CrossRef]
[Pubmed]

20.
Hall-Stoodley L, Stoodley P. Evolving concepts in biofilm infections. Cell Microbiol 2009;11(7):1034–43. [CrossRef]
[Pubmed]

21.
Muschik M, Lück W, Schlenzka D. Implant removal for late-developing infection after instrumented posterior spinal fusion for scoliosis: Reinstrumentation reduces loss of correction. A retrospective analysis of 45 cases. Eur Spine J 2004;13(7):645–51. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Paulien van de Voorde - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Jan AP Geurts - Conception of the work, Design of the work, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guaranter of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestSenior author is member of the BonAlive Clinical Advisory Board.
Copyright© 2019 Paulien van de Voorde et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.